In our latest edition of the CTGCT Science Talks webinar series we had Dr. Thomas Fox, a clinician-scientist whose work bridges the gap between the clinical-laboratory interface working across Harvard Medical School and the University College London. His research focuses on “programmable” T-cell therapies, a field of medicine where we no longer rely solely on external chemicals or drugs to fight disease. Instead, scientists are learning how to rewrite the genetic code of our own immune cells to transform them into “living medicines” capable of seeking out and destroying cancer or correcting rare genetic disorders.
The human immune system is incredibly complex, but it isn’t perfect. Sometimes it lacks the tools to recognize a tumor, and other times it accidentally attacks the body it is supposed to protect. Dr. Fox’s work aims to solve these problems by using gene editing techniques to modify T-cells, an important part of our immune system that has important functions in controlling our immune responses. By combining the precision of CRISPR gene editing with innovative delivery systems like lipid nanoparticles, his team is developing ways to engineer these cells both inside (in vivo) and outside the body.

Fixing the “Brakes” of the Immune System
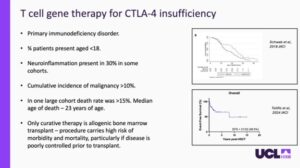
One of the focuses of Dr. Fox’s research is a rare and life-threatening genetic condition known as CTLA4 insufficiency. In a healthy person, the CTLA4 protein acts as a vital regulatory “brake” for the immune system, ensuring that T cells do not become overactive and attack the body’s own healthy tissues. When a patient lacks enough of the CTLA4 protein, their immune system remains in a constant state of high alert, leading to severe inflammation, organ damage, and a significantly higher risk of developing cancer. For many years, the only potential cure for these patients has been a full bone marrow transplant, a high-risk procedure that involves replacing the patient’s entire immune system with a donor’s cells.
To provide a safer and more precise alternative, Dr. Fox and his team are utilizing CRISPR-Cas9 technology to repair the patient’s own T cells. For that they first extract the blood from the patients and genetically modify a specific type of T cells (CD3+ T cells). Rather than simply adding a new gene at random, they use CRISPR technology ( the “molecular scissors” ) to target a specific non-coding region of the DNA called an intron (in particular intron 1). Usually in the process of translating DNA to proteins, this region will be ignored, but by inserting a healthy copy of the CTLA4 gene directly into this natural regulatory site, the cell can use its control machinery to regulate the production of CTL4 protein, avoiding the risks of overproduction. This is a common problem with older gene therapy methods.
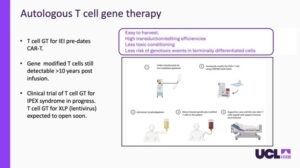
The results of this approach have been highly promising in preclinical studies. When these modified T-cells were injected into the test subjects, they traveled through the body to reach the affected organs and successfully quieted the overactive immune system, helping the swollen and injured tissues heal and return to their proper size (Link to the paper). This breakthrough has paved the way for clinical translation, with a clinical trial scheduled to begin in May 2028, offering hope for a “living drug” that can restore balance to the immune system without the need for a donor transplant.
Stealth T Cells: Epitope Editing
Beyond fixing genetic errors, Dr. Fox explored how to make engineered T cells “stealthy” so they can survive the harsh environment of a patient’s body during treatment. Often, before a patient receives new T cells, doctors use powerful drugs called monoclonal antibodies to clear out the existing, faulty immune cells to make “room” for the new ones. The problem is that these drugs are often too effective; they cannot tell the difference between the “bad” old cells and the “good” engineered ones, meaning the expensive new therapy can be wiped out by the medicine as soon as it is injected.
To solve this, Dr. Fox’s team utilized a technique called epitope editing. An “epitope” is essentially the specific spot or docking site on a protein that an antibody grabs onto to identify a cell. By using CRISPR to change just one or two amino acids in the surface protein of T cells called CD52, the team created a version of the protein that is invisible to the monoclonal antibodies. While other researchers have tried simply deleting the CD52 protein entirely, Dr. Fox discovered that this “knock-out” approach actually made the cells much more vulnerable.

The reason for this lies in the hidden function of the CD52 protein. It does more than just act as a target; it also sends a crucial “don’t eat me” signal to other immune cells called macrophages. When the protein is deleted entirely, the macrophages no longer recognize the T cell as “friendly” and destroy it. By precisely editing the epitope instead of removing the whole protein, the team created “stealth” T cells that are significantly more resilient. These cells are able to hide from the drug while still maintaining their protective signals, making them far more “fit” and effective at surviving to fight disease in the long run.
The “On-Site” Factory: In Vivo Programming
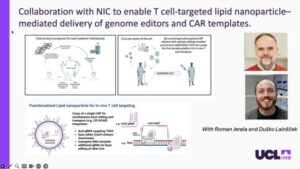
Instead of the weeks-long process of removing and modifying T cells in a lab, Dr. Fox is developing “in vivo” programming to engineer T cells directly inside the patient’s body. In collaboration with Prof. Roman Jerala and Dr. Duško Lainšček from the National Institute of Chemistry and members of our CTGCT team, they used Lipid Nanoparticles (microscopic “fat bubbles”) to deliver genetic instructions to T cells while they are still in the bloodstream.
This approach has shown remarkable potential for bone marrow cancers like leukemia. Because the T cells are programmed right where the cancer is located, they do not have to navigate the difficult journey from the blood into deep bone tissues. Dr. Fox found that these “on-site” cells are naturally hardier and more effective than lab-grown ones, this could lead to turning a complex laboratory manufacturing into a simple, life-saving injection.
Towards a New Era of Living Medicine
Ultimately, in the webinar Dr. Fox showed that the goal of his research is to move beyond the limitations of traditional medicine by creating a new generation of “programmable” therapies. By harnessing the precision of CRISPR-Cas9 and the efficiency of nanoparticle delivery, his work seeks to provide safer, more effective treatments for patients facing aggressive cancers and complex immune disorders. Whether it is fixing a genetic “brake” to stop the body from attacking itself or engineering “stealth” cells that can survive alongside powerful drugs, the objective remains the same: to turn the immune system into a highly controlled and targeted tool for healing.
We would like to thank Dr. Fox for sharing these remarkable insights and for his dedication to pushing the boundaries of what is possible in modern medicine with the CTGCT community. We will keep you updated with more news about our mutual collaboration.
