Imagine if we could talk to our cells the same way we talk to a smart home device. We could tell a cell to start producing a specific medicine only when it’s needed or order a group of cells to shift their behavior once they sense a specific signal in the body. For years, synthetic biologists have been trying to build these “remote controls” for life, but the research presented in this paper from member of the Department of Synthetic Biology and Immunology and the CTGCT at the Kemijski Inštitut has taken this to a whole new level. By using parts already found in our own bodies and a physics trick similar to what makes a lava lamp work, researchers have created a way to turn genes on and off with incredible power and precision.
Key Words and Concepts to Understand
- Chemically Induced Dimerization (CID): This is a biological mechanism in which two proteins bind only in the presence of a certain small molecule, enzyme or other dimerizing agent. Think of this as a molecular “Velcro” system where two protein pieces stay apart until a specific molecule, acting like glue, causes them to stick together.
- Liquid-Liquid Phase Separation (LLPS): This is a similar process to precipitation in a solution, but instead of a solid, similar molecules clump together into droplets that float in the solution. Like the “bubbles” in a lava lamp.
- Biomolecular Condensates: These are the tiny, liquid “bubbles” formed by LLPS inside a cell that allow chemical reactions to happen much faster by crowding parts together.
- Nuclear Receptors: These are natural human proteins that act like antennas or sensors to recognize specific hormones and trigger a response in the cell.
- Transcription: The biological process of “reading” a gene’s DNA instructions to create a copy in RNA that will later be used to create the protein. Think of it as taking a digital photo of a rare library book; you get the information you need in a usable format without risking damage to the original “master” copy.
From Simple Switches to Liquid Power Plants
What makes this research so innovative is the shift from “digital” switches to “amplified” systems. In the past, scientists often used the Nuclear Receptors from bacteria or plants to build these cell controls. While they worked, our immune systems sometimes recognized them as “foreign invaders” and the response they triggered was often quite weak, like a dim lightbulb flickering on. The team of scientists at the Kemijski Inštitut, decided to “repurpose” human receptors instead. They took the sensing portion of human hormone receptors (like those for Vitamin D or Estrogen) and turned them into a high-tech Chemically Induced Dimerization (CID) system. Because these parts are human-derived, they are much more likely to be safe for use in medical treatments without causing an immune reaction.
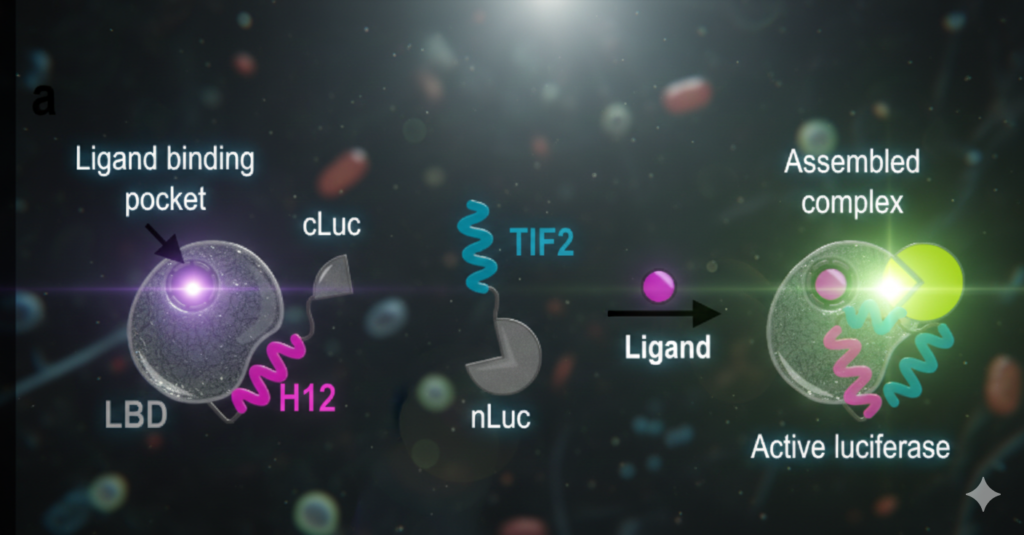
The real “magic” happened when they combined these human receptors with the concept of liquid droplets. By designing Nuclear Receptors with multiple “sticky” ends (known as ligand binding domains), the team found that once they added a hormone, the proteins wouldn’t just pair up; they would undergo Phase Separation.
For example, when a hormone like cortisol or estrogen (the ligand) is introduced, it binds to the Ligand Binding Domain (sticky end) of the Nuclear Receptor. This causes the Ligand Binding Domain to undergo a structural change, specifically moving a part called Helix 12, to reveal a “hydrophobic groove”, a groove in the surface of the Nuclear Receptor. This groove provides the perfect physical environment for the other peptide in this dimer, a peptide called TIF2, to then “stick” to the Nuclear Receptor.
The innovation here is that scientists used multiple repeats of these TIF2 peptides (up to six in a row) and clustered the ligand binding domains together. Because there are so many sticky points available at once, the proteins don’t just find one partner and stop. Instead, they link with each other rapidly, causing them to “exit” of the surrounding cellular fluid and clump together into these dense liquid droplets, the Biomolecular Condensates. This is the Liquid -Liquid Phase Separation process we explained above, which effectively creates a concentrated “reaction hub bubble” that amplifies the gene-triggering signal by hundreds of times.
This causes thousands of gene-activating molecules to suddenly rush into a single, dense liquid droplet right on top of the DNA. This created a “threshold effect.” Instead of a slow, steady increase in activity, the gene expression stayed low until enough hormones were present to form a droplet, at which point the activity “exploded” to several hundred times its original level. This provides a level of power and “gain” that was previously very difficult to achieve in synthetic biology.
Why This Research Matters for Our Future
The implications of this work for medicine and biotechnology could be profound. Because the researchers used receptors that respond to common, clinically approved drugs and natural hormones like cortisol or estrogen, we already have the “remote controls” sitting in our medicine cabinets. This could lead to a new generation of “smart” cell therapies. For example, a patient could have engineered cells that stay dormant until they take a specific, safe pill that triggers these liquid droplets to form, turning on a powerful therapeutic response only when and where it is needed.
Furthermore, the system is completely reversible. By adding a different molecule, known as an “antagonist” that blocks the Nuclear Receptor, the researchers showed they could dissolve the liquid droplets and turn the gene back off almost instantly. This “kill switch” is vital for safety in medicine. By integrating synthetic biology with the body’s own natural signaling systems, it could prove critical in the design of future treatments that will not only be more powerful but will be more in tune with our immune system.